Beta-Glucans Market Forecast 2026–2036: Market to Reach USD 3.0 Billion at 5.8% CAGR
Beta-Glucans Market is segmented by Source (Yeast, Oats, Barley, Mushrooms, and Others), Functionality (Soluble Beta-Glucan and Insoluble Beta-Glucan)
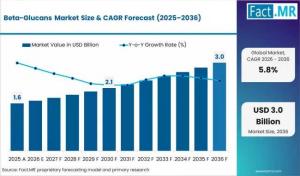
ROCKVILLE, MD, UNITED STATES, March 18, 2026 /EINPresswire.com/ -- As global healthcare shifts from reactive treatment to proactive wellness, one specific class of polysaccharides is emerging as the gold standard for clinical-grade efficacy: Beta-glucans. Valued at USD 1.7 billion in 2026, the global market is on an aggressive trajectory to reach USD 3.0 billion by 2036, according to the latest strategic analysis. This represents a staggering USD 1.3 billion in new revenue opportunities over the next decade as the ingredient moves from a simple "fiber" classification to a highly sought-after bioactive agent.Get Access Report Sample : https://www.factmr.com/connectus/sample?flag=S&rep_id=14559
The Regulatory Renaissance: From Commodity to Clinical Grade
The primary catalyst for this expansion is a structural shift in how ingredients are marketed to a more sophisticated consumer base. The market is currently pivoting away from commodity dietary fiber toward specialized, characterized beta-glucans supported by rigorous clinical evidence.
A "Regulatory Renaissance" is taking hold across key markets:
Japan’s FOSHU Evolution: Recent expansions in FOSHU (Food for Specified Health Uses) approvals have enabled premium beta-glucan products to command 30–50% price premiums.
European EFSA Mandates: In Germany and the Netherlands, EU Regulation 432/2012 has institutionalized cholesterol-lowering claims, forcing a mass-market reformulation of cereals and bakery products.
North American Clinical Integration: In the United States, the FDA’s updated 2026 health claims are bridging the gap between oat-based heart health and yeast-derived immune support, particularly within hospital dietary programs.
The Yeast vs. Oat Tug-of-War: Segmental Dynamics
While the market is diverse, two sources are battling for dominance in the 2026 landscape. Yeast-derived beta-glucans are projected to hold a 30% market share, favored by pharmaceutical-grade supplement formulators for their immune-modulating properties. Companies like Lesaffre Group have recently expanded production capacity to meet this demand, while Biothera Pharmaceuticals has released clinical data further validating respiratory health benefits.
Conversely, soluble beta-glucans—primarily from oats and barley—account for 45% of the market functionality share. This is driven by their unique viscosity-forming mechanisms that satisfy the strict 3g daily dose requirements for cholesterol-reduction claims. Innovation in extraction, such as Kerry Group’s high-concentration processes and Tate & Lyle’s improved cold-water dispersibility, is making it easier for manufacturers to fortify beverages and dairy without compromising texture.
Regional Powerhouses: Japan and Australia Lead Growth
Growth trajectories are being written by regional regulatory maturity and aging demographics:
Japan (7.0% CAGR): The fastest-growing market, where elderly populations prioritize immune support, leading to five new FOSHU applications for yeast beta-glucan in the last year alone.
Australia (6.7% CAGR): Driven by the TGA’s "Listed Medicine" pathway, which has allowed brands like Blackmores and Swisse to dominate the pharmacy-based immune supplement category.
Canada (6.0% CAGR): Bolstered by Health Canada’s heart-health claims, which supported a wave of new Natural Health Product (NHP) licenses in 2026.
Competitive Landscape: The Battle for "Sticky" Formulations
The global beta-glucans market remains moderately fragmented, but the advantage is shifting toward vertically integrated players who can provide clinical-grade "proof of work."
Key Industry Participants include:
Kemin Industries Inc., Lesaffre Group, Biothera Pharmaceuticals Inc., Ceapro Inc., Garuda International Inc., Kerry Group Plc, Tate and Lyle Plc, Biotec BetaGlucans AS, Super Beta Glucan Inc., and Angel Yeast Co., Ltd.
Success in this sector is no longer about volume; it is about regulatory expertise. Suppliers that can align their clinical trials with the specific labeling requirements of the FDA, EFSA, and FOSHU are securing long-term contracts with functional food giants and clinical nutrition programs.
Analyst Insights: The Strategic Outlook
"FOSHU approvals, EFSA claims, and TGA-listed supplements are creating premium, regulatory-driven markets that support significantly higher margins for clinical-grade producers," says Shambhu Nath Jha, Principal Consultant at Fact.MR. "The winners in this space will be the manufacturers who stop fighting the variation in raw materials and start standardizing the clinical outcomes for the end-user."
Conclusion: A Decade of Bioactive Integration
Looking toward 2036, beta-glucans are set to become a ubiquitous component of the "smart" pantry and the clinical medicine cabinet. As the cosmetics industry further adopts these polysaccharides for skin-barrier and microbiome support, the market's reach will extend into every facet of the wellness economy. For stakeholders, the message is clear: the future of beta-glucans lies in the intersection of traceable supply chains and ironclad clinical validation.
Browse Full Report – https://www.factmr.com/report/beta-glucans-market
To View Related Report:
Organic Cocoa Market https://www.factmr.com/report/organic-cocoa-market
Fortified Foods Market https://www.factmr.com/report/fortified-foods-market
Erythritol Market https://www.factmr.com/report/erythritol-market
Citrus Flavor Market https://www.factmr.com/report/citrus-flavor-market
S. N. Jha
Fact.MR
+1 628-251-1583
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.